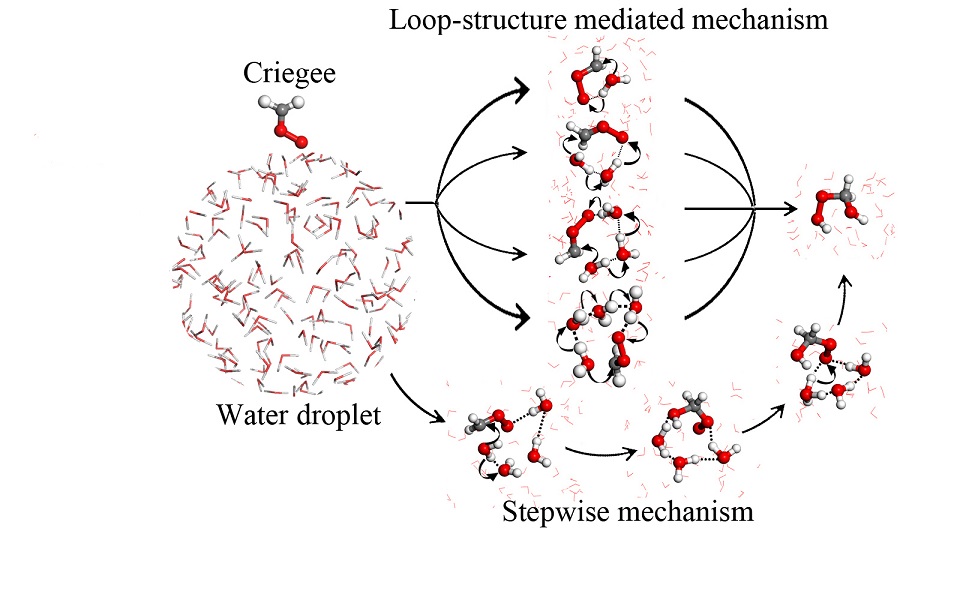
Alkenes constitute a large fraction of the natural and human-made volatile organic compounds (VOCs) that are emitted into the troposphere. Their oxidation products degrade air quality and contribute to climate warming. Alkene oxidation is thought to involve Criegee intermediates (CIs), highly reactive molecules that form when ozone reacts with alkenes. Early experimental studies showed that the Criegee intermediate did not seem to react with water. However, modelers have found it difficult to quantify the effect of CIs on atmospheric composition, because laboratory data on CI reactions with water have been contradictory. A later study by Chao et al. (Science, DOI: 10.1126/science.1261549) showed that the simplest Criegee intermediate, CH2OO, does in fact react very rapidly with pairs of water molecules. The new rate measurements imply that reactivity with atmospheric water pairs is a major decay channel for Criegees. Given that 65% of the Earth is covered by clouds, no studies have given attention to the effect of clouds or aerosols on Criegee chemistry. New NC3 study (J. Am. Chem. Soc., DOI: 10.1021/jacs.6b04338 ) now indicates that the time scale for the reaction of CH2OO with water at the air/water interface is 2-3 orders of magnitude faster than that in the gas phase, suggesting that clouds should be a major sink for the Criegee intermediate.



