Charles Mach University Professor

University of Nebraska-Lincoln
807B Hamilton Hall
Lincoln, NE 68588-0304
(402) 472-6232
jtakacs1@unl.edu
Education
Postdoctoral, Swiss National Funds International Postdoctoral Fellow, Swiss Federal Institute of Technology
Ph.D., California Institute of Technology
B.S., Rutgers University
Research Interests
Asymmetric Catalysis, Self-Assembly, Combinatorial Catalysis, Metal-Catalyzed Cyclizations, Synthetic Methods Development, Natural Product Total Synthesis
Current Research
Catalytic Asymmetric Hydroboration (CAHB) has been around for a long time, and many outstanding research groups have contributed to understanding this (fiendishly complex) catalyzed counterpart of the classic stoichiometric reaction.[i] Now, as new methods rapidly evolve using chiral secondary and tertiary boronic esters for stereospecific carbon-carbon bond construction,[ii] the development of new catalyst systems and the exploration of new substrates for CAHB are enjoying renewed interest. Recent work in the Takacs group has focused on developing the CAHB, mostly rhodium-catalyzed CAHBs, of substrates capable of two-point binding to the metal center. The observation of unusual Markovnikov regioselectivity leading to chiral tertiary boronic esters is among the important advances. For example, we recently published a simple, in situ-generated, chiral rhodium catalyst for which methylidene and trisubstituted alkene substrates form chiral tertiary boronic esters (up to 87% yield and 96:4 enantiomer ratio (er)) via oxime-directed CAHB. Subsequent C–C coupling is used to form a quaternary all carbon stereocenter, while other transformations lead to chiral diols, O-substituted hydroxylamines, and isoxazolines.[iii] Similarly, as illustrated below, phosphonate-derived CAHB affords new, bifunctional chiral tertiary boronic ester synthons suitable for further elaboration.[iv] Recently, it has been discovered that H2 will “hitch a ride” on the CAHB catalyst interrupting hydroboration to arrive at efficient Catalytic Asymmetric Hydrogenation (CAH).[v]
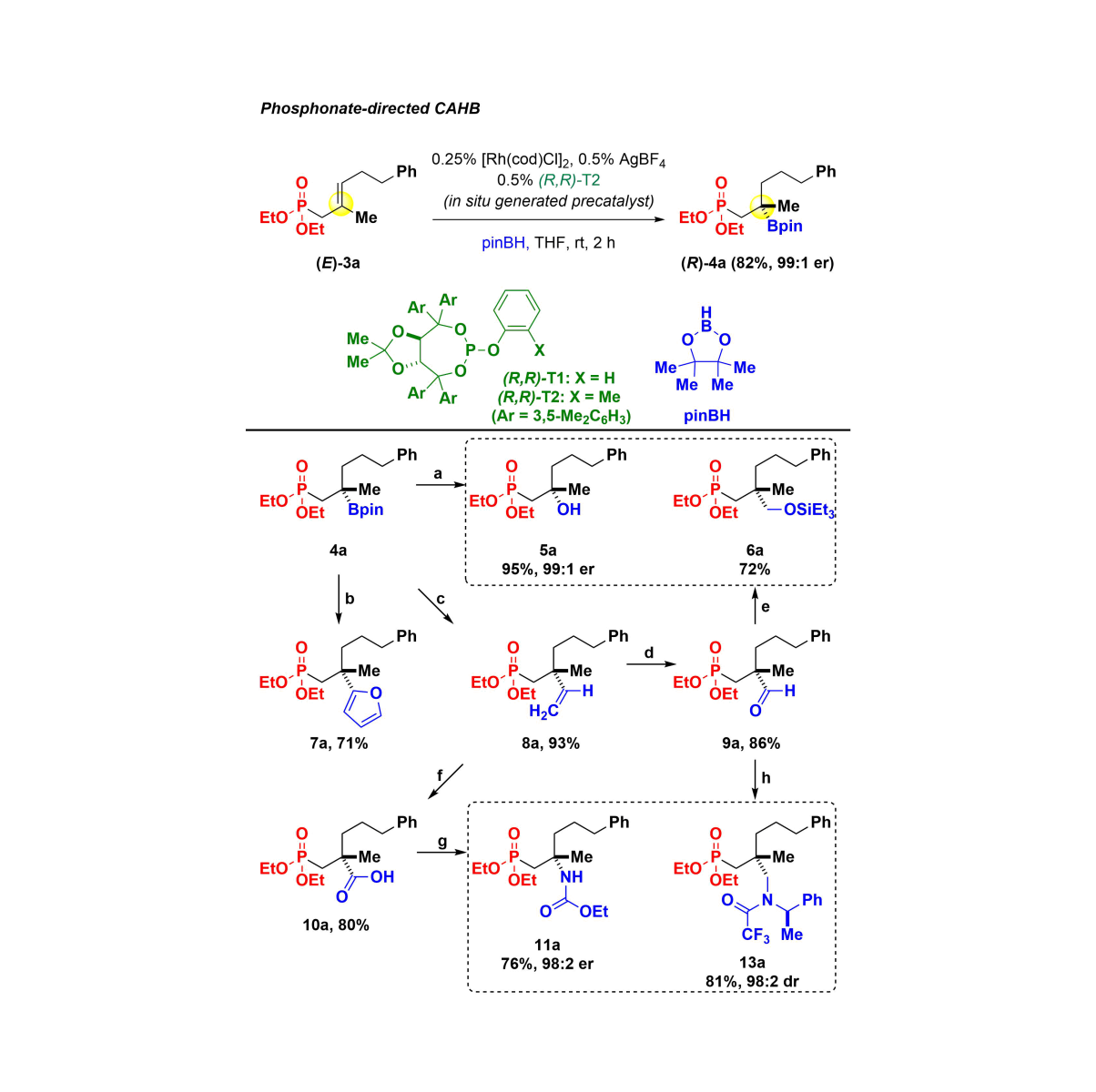
[i] A. M. Carroll, T. P. O’Sullivan, P. J. Guiry, Adv. Synth. Catal., 2005, 347, 609–631; b) C. M. Crudden, D. Edwards, Eur. J. Org. Chem., 2003, 24, 4695–4712.