.png)
Assistant Professor Jian Zhang and his colleagues have synthesized a new class of highly porous and stable metal-organic frameworks (MOFs) with a novel topology. In their recent work, they described the assembly of elongated tetrahedral linkers with stable zirconium-oxo clusters to construct two non-interpenetrated Zr-MOFs, NPF-200 and NPF-201 (NPF: Nebraska Porous Framework). Upon activation, NPF-200 exhibits the largest BET surface area (5,463 m2 g–1) among all MOFs formed from tetrahedral ligands.
Metal-organic frameworks (MOFs) is a class of porous crystalline materials that has witnessed the rapid development of the past two decades.1 The tunability of both organic linkers and inorganic metal cluster based secondary building units (SBUs), combined with large surface areas and convenient chemical modification methods, have enabled their potential applications in gas storage and separation, catalysis, chemical sensing, drug delivery, among others.2 However, many MOFs are water/moisture-sensitive due to the reversible coordination bonding, considerable effort has been focused on MOFs with high thermal and chemical stability for their ultimate practical applications. Zirconium-based MOFs (Zr-MOFs) represent a remarkable improvement to this end: the high valence of ZrIV and large ZrIV-O bond polarization can lead to a strong bonding between ZrIV and carboxylate oxygens in the ligands.3 Nevertheless, this specific type of MOFs is still in its early stage of development. Its wider practical applications call for continuous discovery of new structures with novel topologies, which is important to understanding the fundamental correlation between framework structure and porosity, stability, and interpenetration.
Zhang and co-workers overcame the synthetic challenge and prepared two elongated tetrahedral linkers. Followed by a solvothermal reaction of ZrCl4 with the linkers in N,N-dimethylformamide (DMF), single crystals of NPF-200 and 201 were successfully obtained. X-ray diffraction study on the reveals NPF-200 a non-interpenetrated framework that is composed of two different types of 12-connected Zr clusters (Zr6 and Zr8) linked by tetrahedral ligands (Figure 1a). Overall, it is a trinodal net with the 4,12,12T1 topology, which is the same with that of β-UH3 (Figure 1b).4 It is noteworthy that NPF-200 represents the first MOF structure that exhibits this rare topology. The Zr6 and Zr8 clusters in NPF-200 can be envisioned as the topological close packing phase A15 (Figure 1c),5 one of the Frank-Kasper phases used to describe the structures of intermetallics with A3B stoichiometry.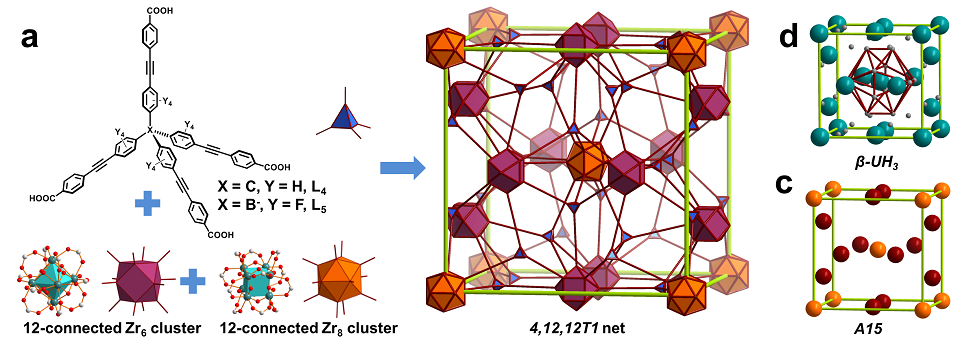
NPF-200 has the N2 uptake at 77K of 1403 cm3 g–1, the SABET (Brunauer-Emmett-Teller surface area) of 5463 m2 g–1 (Figure 2a), and the SALangmuir (Langmuir surface area) of 6877 m2 g–1. The solvent accessible volume of NPF-200 is 81.6% with a pore volume of 2.17 cm3 g–1%. It is noted that NPF-200 has the largest surface area, pore size, and solvent-accessible volume among all MOFs constructed from tetrahedral ligands. The calculated volumetric surface area of NPF-200 is 2125 m2 cm–3, which is outstanding among many well-known MOFs. Remarkably, NPF-200 shows high stability in acidic and basic conditions (Figure 2b). After soaking in aqueous solutions with pH values ranging from 1 to 11 for 24h, the PXRD patterns of NPF-200 are almost unchanged, suggesting the preserved crystallinity (Figure 2b).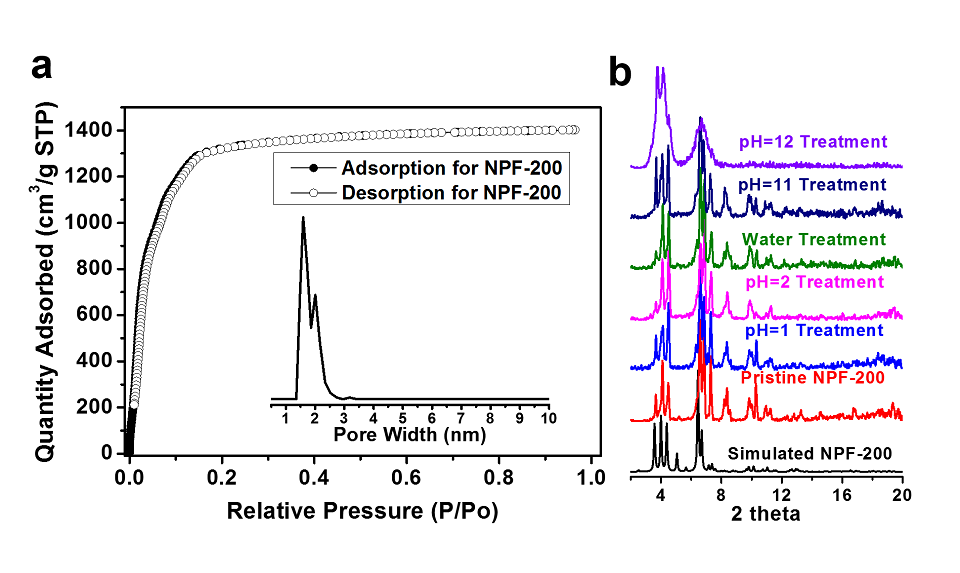
The team then synthesized NPF-201 as an isostructure of NPF-200 using a boron-centered tetrahedral linker and obtained the resulting anionic Zr-MOF. Photoactive cationic transition metal complex, [Ru(bpy)3]2+, was incorporated into NPF-201 via ion exchange to form Ru@NPF-201, which exhibits the characteristic emission band of [Ru(bpy)3]2+ at 600 nm and was used as a heterogeneous catalyst in photo-oxidation of thioanisole. Overall, Zhang group’s work has pointed a new direction for designing functional Zr-MOFs with new topologies and potential new applications in gas storage and catalysis.
The study is published in JACS and is viewable at https://pubs.acs.org/doi/abs/10.1021/jacs.6b04608
Reference:
(1) Furukawa, H.; Cordova, K. E.; O'Keeffe, M.; Yaghi, O. M. Science 2013, 341, 1230444.
(2) Suh, M. P.; Park, H. J.; Prasad, T. K.; Lim, D. W. Chem. Rev. 2012, 112, 782-835.
(3) Bai, Y.; Dou, Y.; Xie, L. H.; Rutledge, W.; Li, J. R.; Zhou, H. C. Chem. Soc. Rev. 2016, 45, 2327-2367.
(4) Rundle, R. E. J. Am. Chem. Soc. 1951, 73, 4172-4174.
(5) Graef, M. D.; Mchenry, M. E. Structure of Materials: An Introduction to Crystallography, Diffraction and Symmetry; 2 ed.; Cambridge Univ. Press: Cambridge, 2012.



