
University of Nebraska-Lincoln
704 Hamilton Hall
Lincoln, NE 68588-0304
(402) 472-2744
dhage1@unl.edu
Education
Postdoctoral, Mayo Clinic
Ph.D., Iowa State University
B.S., University of Wisconsin
Research Interests
Analytical chemistry, bioanalytical chemistry, analytical separation methods, high-performance liquid chromatography, capillary electrophoresis, affinity-based separations, affinity mass spectrometry, monolithic supports and hybrid separation media, affinity microcolumns and miniaturized separation systems, separation-based studies of biological interactions, analytical methods for personalized medicine, chromatographic immunoassays, immobilization methods for biochemicals
Current Research
Our laboratory uses biological ligands in high performance liquid chromatography (HPLC) and capillary electrophoresis (CE) for the analysis of clinical, pharmaceutical, and environmental agents. One area of our work is in the use of antibodies and antibody mimics to create rapid chromatographic-based immunoassays. An example is a series of new methods we have recently created for the measuring the free, or biologically-active, fraction of drugs and hormones in blood or serum. These methods make use of antibodies with special microcolumns and supports that allow these separations to be performed in milliseconds. Similar work has been performed to create selective extraction systems and field-portable devices to isolate and analyze environmental contaminants in water.
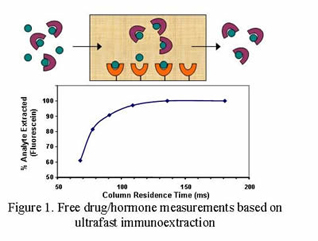
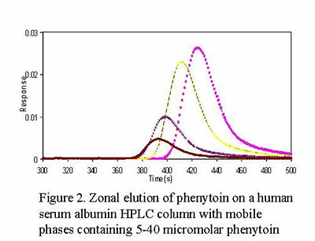
A second area of research in our group is in the use of separation methods to study biological interactions. For example, we use immobilized proteins in HPLC columns to examine the equilibrium constants and rate constants for these proteins as they bind to drugs or other proteins in the body. This information is used to help us develop better analytical methods for these proteins and drugs or to help characterize how such agents behave in the body. The same information can be used to help develop and optimize HPLC and CE systems that employ proteins as binding agents for chiral separations.
We use various tools along with HPLC and CE to help us in our work. Part of this involves the development of new strategies for immobilizing biological ligands like proteins onto solid supports for use in HPLC systems. We are also interested in using new types of supports for this purpose, with both silica-based materials and monolithic columns being employed in our work. Various spectroscopic methods are used to help us characterize these materials, including absorbance spectroscopy, fluorescence spectroscopy, and mass spectrometry. Computer modeling and work with chromatographic theory is also utilized in our group to help us better understand the separation methods we are creating and to design new formats for these methods.
Center for Nanohybrid Functional Materials (CNFM): We are part of the Center for Nanohybrid Functional Materials, a group of fourteen investigators from UNL Chemistry, UNL Electrical Engineering, and four other Nebraska colleges or universities. The activities of the Center focus on interdisciplinary approaches to discovery and application of new sensing and separation principles at the surfaces of functionalized nanomaterials. Our research in this area involves the production of hybrid separation media or new separation media that use nanomaterials, the evaluation of nanomaterials by separation-based methods, and the creation of flow-based bioassays based on nanomaterials and miniaturized separation devices.

For more information, please visit the Hage Research Group Homepage.
Selected Publications
(1) Sobansky, M.R.; Hage, D.S. "Identification and analysis of stereoselective drug interactions with low density lipoprotein by high-performance affinity chromatography", Anal. Biochem. Chem. 2012;403;563-71.
(2) Matsuda, R.; Anguizola, J.; Joseph, K.S.; Hage, D.S. "High-performance affinity chromatography and the analysis of drug interactions with modified proteins: binding of gliclazide with glycated human serum albumin", Anal. Bioanal. Chem. 2011;401;2811-19.
(3) Barnaby, O.S.; Cerny, R.L.; Clarke, W.; Hage, D.S. "Quantitative analysis of glycation patterns in human serum albumin Using 16O/18O-labeling and MALDI-TOF MS", Clin. Chim. Acta 2011;412;1606-15.
(4) Tong, Z.; Hage, D.S. "Characterization of interaction kinetics between chiral solutes and human serum albumin by using high-performance affinity chromatography and peak profiling", J. Chromatogr. A 2011;1218;6892-7.
(5) Hage, D.S.; Anguizola, J.A.; Jackson, A.J.; Matsuda, R.; Papastavros, E.; Pfaunmiller, E.; Tong, Z.; Vargas-Badilla, J.; Yoo, M.J.; Zheng, X. "Chromatographic analysis of drug interactions in the serum proteome", Anal. Methods 2011;3;1449-60.
(6) Yoo, M.J.; Hage, D.S. "High-throughput analysis of drug dissociation from serum proteins using affinity silica monoliths", J. Sep. Sci. 2011;34;2255-63.
Complete list of publications