
University of Nebraska-Lincoln
524 Hamilton Hall
Lincoln, NE 68588-0304
(402) 472-2734
ceckhardt1@unl.edu
Education
Ph.D., Yale University
M.S., Yale University
B.A., University of Colorado
Research Interests
Solid-state reactions; electronic and vibrational spectroscopy of molecular crystals; lattic dynamics; modluation spectroscopy; crystal engineering
Current Research
The role of mechanical energy in chemical processes is being addressed through determining the mechanical properties of energetic materials and pharmaceuticals, both notoriously susceptible to stress-induced chemistry. Of particular interest is how chemical behavior is related to mechanical stress induced either by pressure, defects or light. Understanding requires quantitative measure of the anisotropy of elasticity, the nature and concentration of defects and the photoelasticity of various organic materials. This information is virtually non-existent and is being used to provide a foundation for understanding processes such as detonation and "browning" of pharmaceuticals.
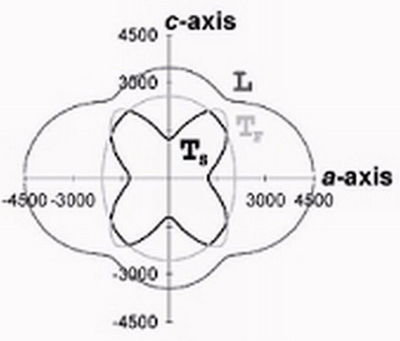
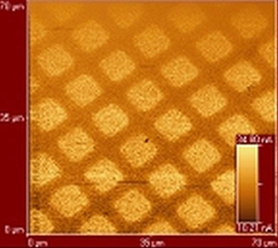
The microscopic origins of friction are being investigated by design of unique amphiphiles that are used to form either Langmuir-Blodgett or SAM films. The surfaces formed by the films have specific chemical modifications that are designed to influence the frictional properties of the films. The nanofriction, measured friction force microscopy, is then compared to macroscopic friction.
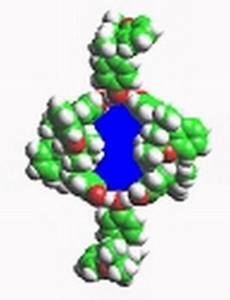
Intermolecular forces are being studied through the guest-host interactions of inclusion compounds. The influence of different guests on the physical properties, e.g. elasticity, heat of fusion, of the host crystals is related to intermolecular interactions.
For more information, please visit the Eckhardt Research Group Homepage.
Selected Publications
(1) Eckhardt, C. J.. Organic Molecular Solids. Molecular Crystals and Liquid Crystals (2008), 487 173-174. CODEN: MCLCD8 ISSN:1542-1406. AN 2008:986515 CAPLUS
(2) Mohapatra, Himansu; Eckhardt, Craig J.. Elastic Constants and Related Mechanical Properties of the Monoclinic Polymorph of the Carbamazepine Molecular Crystal. Journal of Physical Chemistry B (2008), 112(8), 2293-2298.
(3) Eckhardt, Craig J.; Gavezzotti, Angelo. Computer Simulations and Analysis of Structural and Energetic Features of Some Crystalline Energetic Materials. Journal of Physical Chemistry B (2007), 111(13), 3430-3437.