
University of Nebraska-Lincoln
634AA Hamilton Hall
Lincoln, NE 68588-0304
(402) 472-3525
jguo4@unl.edu
Education
Postdoctoral, The Scripps Research Institute
Ph.D., Michigan State University
B.S. & M.S., Nankai University
Research Interests
Chemical biology, bioorganic chemistry, and synthetic biology.
For more information, please visit the Guo Research Group Homepage.
Current Research
The Guo group is a place where chemistry meets biology. Our group uses a multidisciplinary approach to tackle challenges associated with human health and sustainable chemical production. We seek to translate scientific discoveries into biomedical and biotechnological applications.
Our research focuses on the elucidation of molecular mechanisms that are used by living organisms to achieve essential biological and/or chemical transformations, and on the development of enabling methodologies for both biochemical investigations and biomedical applications. We are also engaged in the use of Synthetic Biology approach to re-write or re-wire genetic information so that a cell or an organism can be programmed to perform a pre-defined task.
A graduate student or postdoctoral researcher in my lab can expect to receive training in bioorganic chemistry, chemical biology, and synthetic biology, and significant exposure to one or more of the following specialties: organic synthesis, enzymatic synthesis, combinatorial (bio)chemistry, bio-molecular engineering, directed biomolecular evolution, unnatural amino acid mutagenesis, protein biochemistry, and molecular biology.
For more information, please visit the Guo Research Group Homepage.
Selected Publications
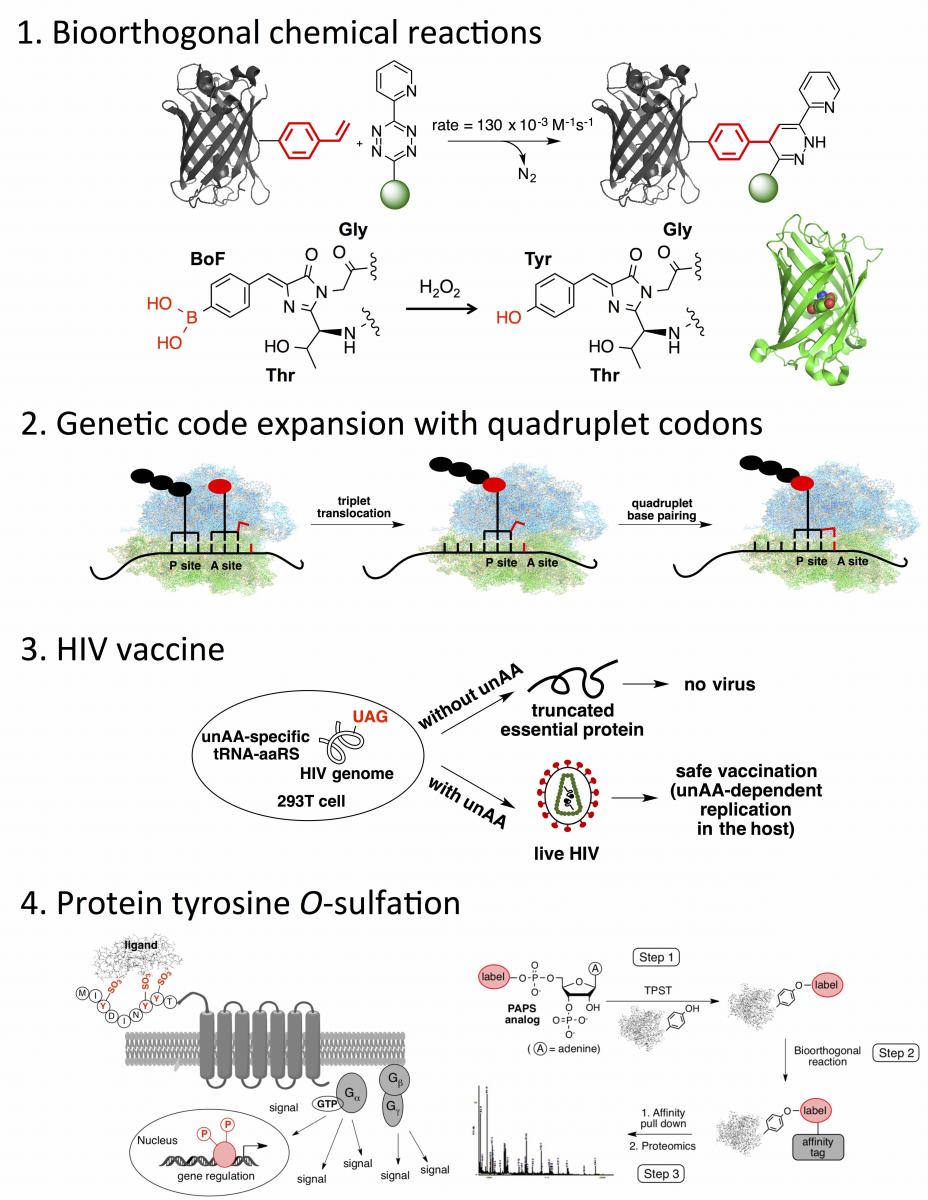
(1) Liu, K.; Enns, B.; Evans, B.; Wang, N.; Shang, X.; Sittiwong, W.; Dussault, P.;* Guo, J.* “A Genetically Encoded Cyclobutene Probe for Labeling of Live Cells” Chem. Commun. 2017, 53, 10604-10607. [Link]
(2) Shang, X.; Wang, N.; Cerny, R.; Niu, W.; Guo, J.* “Fluorescent Protein-Based Turn-On Probe Through A General Protection-Deprotection Design Strategy” ACS Sens. 2017, 2, 961–966. [Link]
(3) Shang, X.; Song, X.; Faller, C.; Lai, R.; Li, H.; Cerny, R.; Niu, W.; Guo, J.* “Fluorogenic Protein Labeling Using A Genetically Encoded Unstrained Alkene” Chem. Sci. 2017, 8, 1141-1145. [Link]
(4) Niu, W.*; Guo, J.* “Stereospecific Microbial Conversion of Lactic Acid into 1,2-Propanediol” ACS Synth. Biol. 2015, 4, 378-382. [Link]
(5) Wang, N.; Li, Y.; Niu, W.; Sun, M.; Cerny, R.; Li, Q.;* Guo, J.* “Construction of a Live-Attenuated HIV-1 Vaccine through Genetic Code Expansion” Angew. Chem. Int. Ed. 2014, 126, 4967-4971. ( This article was highlighted in Nature Chemistry.) [Link]
(6) Niu, W.; Schultz, P. G.; Guo, J.* “An Expanded Genetic Code In Mammalian Cells With a Functional Quadruplet Codon” ACS Chem. Biol. 2013, 8, 1640-1645. (the second most read article in May, 2013; podcast interview with the journal) [Link]
(7) Wang, F.; Niu, W.; Guo, J.*; Schultz, P. G.* “Unnatural Amino Acid Mutagenesis of Fluorescent Proteins” Angew. Chem. Int. Ed. 2012, 51, 10132-10135. [Link]
(8) Guo, J.; Wang, J.; Anderson, J. C.; Schultz, P. G. “Addition of an a-Hydroxy Acid to the Genetic Code of Bacteria” Angew. Chem. Int. Ed. 2008, 47, 722 –725. (This article was highlighted in Nature.) [Link]