
University of Nebraska-Lincoln
824A Hamilton Hall
Lincoln, NE 68588-0304
(402) 472-2738
dberkowitz1@unl.edu
Education
Merck Postdoctoral Fellowship, Yale University
Ph.D., Harvard University
B.S., University of Chicago
National/International Leadership
Division Director, NSF Division of Chemistry (2020)
Co-Chair, Gordon Research Conference on Biocatalysis (2018)
Awards/Honors
2016 - Board of Editors - Organic Reactions
2015 - Fellow, American Association for the Advancement of Science (AAAS)
2010 - Inaugural WCCAS, Kickoff Plenary Lecturer, Beijing, China
2008 - Japan Society for the Promotion of Science Fellow
2006 - Visiting Professor, Max Planck Institute for Molecular Physiology, Dortmund, Germany
2005 - Visiting Professor, U. de Rouen, Rouen, France
1997-2001 - Alfred P. Sloan Fellow
Research Interests
Catalyst screening and reaction development, asymmetric synthesis, fluorinated phosphonates, mechanism-based enzyme inhibitors, PLP enzymes, lignan natural products
Current Research
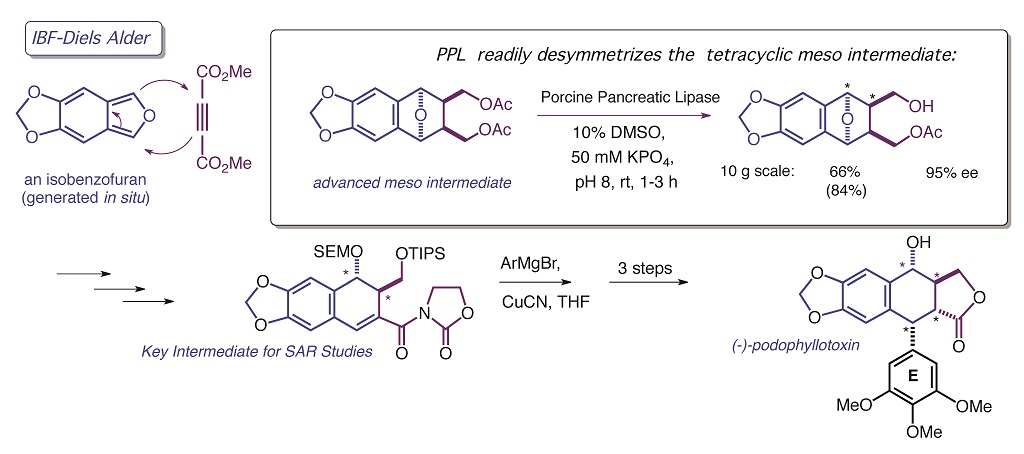
Our group uses the power of stereocontrolled organic synthesis to address questions in biological chemistry, particularly those related to protein-ligand interactions. For example, Fig. 1 illustrates the first catalytic, asymmetric synthesis of (-)-podophyllotoxin, which serves as a tool for us to examine how the structure of the E-ring affects drug binding to tubulin. Total synthesis has yielded compounds more potent than the natural product itself, both in the tubulin assay and against human cancer cell lines.

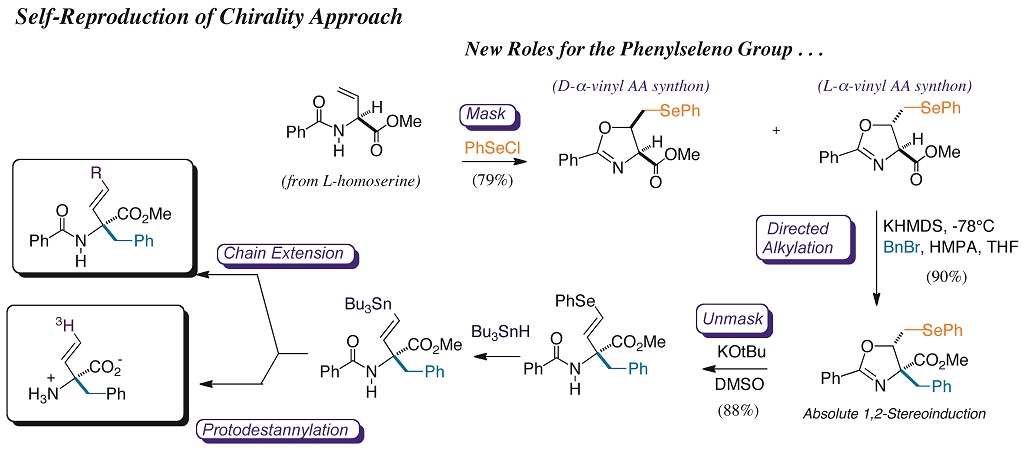
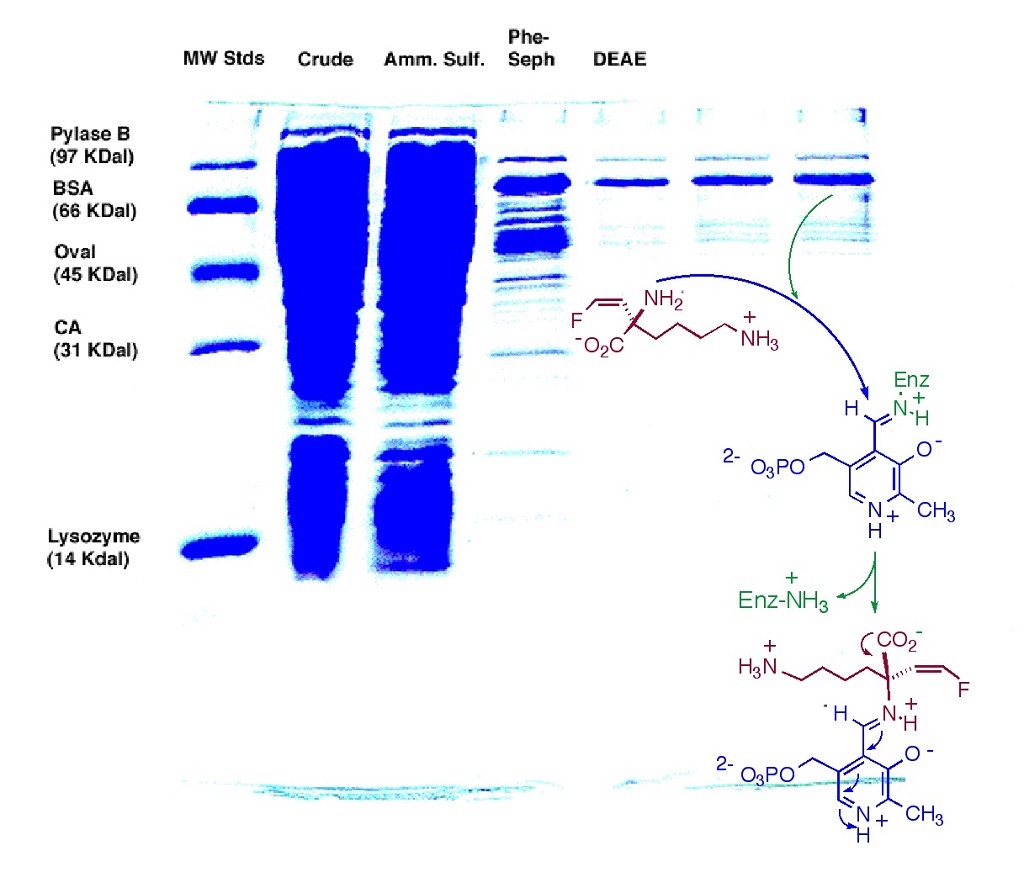
We are also engaged in the synthesis and evaluation of unnatural analogues of amino acids (e.g. Fig. 2), designed to inactivate target enzymes. Coworkers on this project learn protein purification skills (Fig. 3), as well as enzyme kinetics, to characterize the nature of the inactivation. In a complementary endeavor, we construct mimics of natural phosphate esters that are inert to ubiquitous, digestive phosphatase enzymes. We use these phosphate mimics as bioorganic tools to build unnatural ligands for important natural phosphate binding pockets in enzymes (e.g. glucose 6-phosphate dehydrogenase) or receptors (e.g. M6P-IGF2R). Our phosphoserine mimic has served as an important bioorganic tool for biomedical scientists, at the NIH and Johns Hopkins, respectively, to study signal transduction in human tumor suppression (p53 pathway), and in production of the time-keeping hormone, melatonin.
In an exciting new development, we have turned the tables, and use enzymes to assist organic chemists in inventing new reactions through combinatorial catalysis. Arrays of potential catalysts are screened with "reporting" enzymes to provide the chemist with on the fly information about catalyst rate and enantioselectivity. We term this approach ISES (In Situ Enzymatic Screening).
For more information, please visit the Berkowitz Research Group Homepage.
Selected Publications
(1) Robert A. Swyka & David B. Berkowitz “The In Situ Enzymatic Screening (ISES) Approach to Reaction Discovery and Catalyst Identification” Current Protocols in Chemical Biology, 2017, 9(4), 285-305; DOI: 10.1002/cpch.30
(2) Guillaume Malik, Rob Swyka, Virendra Tiwari, Xiang Fei, Greg Applegate, and David B. Berkowitz “A thiocyanopalladation/carbocyclization transformation identified through enzymatic screening: stereocontrolled tandem C–SCN and C–C bond formation” Chem. Sci., 2017, Advance Article; DOI: 10.1039/C7SC04083K
(3) Christopher D. McCune, Matthew L. Beio, Jill M Sturdivant, Roberto de la Salud-Bea, Brendan M. Darnell, and David B. Berkowitz “Synthesis and Deployment of an Elusive Fluorovinyl Cation Equivalent: Access to Quaternary α-(1’-Fluoro)vinyl Amino Acids as Potential PLP Enzyme Inactivators” JACS, 2017, 139, 14077-14089; DOI: 10.1021/jacs.7b04690
(4) Xiang Fei; Megan E. Zavorka, Guillaume Malik, Christopher M. Connelly, Richard G. MacDonald,* David B. Berkowitz,* “General Linker Diversification Approach to Bivalent Ligand Assembly: Generation of an Array of Ligands for the Cation Independent Mannose 6-Phosphate Receptor (CI-MPR).” Organic Letters, 2017, 19, 4267-4270; DOI: 10.1021/acs.orglett.7b01914
(5) David L. Nelson; Gregory A. Applegate, Matthew L. Beio, Danielle L. Graham, David B. Berkowitz,* “Human Serine Racemase Structure/Activity Relationshp Studies Provide Mechanistic Insight and Point to Position-84 Base as a Hotspot for beta-Elimination Function.” J. Biol. Chem. 2017, 292, 13986-14002; DOI: 10.1074/jbc.M117.777904
(6) Christopher D. McCune; Su Jing Chan; Matthew; Beio; Weijun Shen; Woo Jin Chung,; Laura Szczesniak; Chou Chai; Shu Qing Koh , Peter T.-H. Wong;* David Berkowitz* “Zipped Synthesis” by Cross-Metathesis Provides a CBS (Cystathionine β-Synthase) Inhibitor that Attenuates Cellular H2S Levels and Reduces Neuronal Infarction in a Rat Ischemic Stroke Model," ACS Central Science 2016, ASAP; DOI: 10.1021/acscentsci.6b00019; featured in Science Daily: https://www.sciencedaily.com/releases/2016/03/160309082748.htm; featured in Medical News Today: https://www.medicalnewstoday.com/releases/307731.php
(7) Kannan R. Karukurichi, Xiang Fei, Robert A. Swyka, Sylvain Broussy, Sangeeta Dey, Weijun Shen, Sandip K. Roy, David B. Berkowitz* “Mini-ISES Identifies Promising (Carba)fructopyranose-Based Salens for Asymmetric Catalysis: Tuning Ligand Shape via the Anomeric Effect.” Science Advances 2015, 1(6), e1500066; DOI: 10.1126/sciadv.1500066
(8) Gregory A. Applegate and David B. Berkowitz* “Exploiting Dynamic Reductive Kinetic Resolution (DYRKR) in Stereocontrolled Synthesis” Advanced Synthesis & Catalysis 2015, 357, 1619-1632, chosen as a VIP (Very Important Publication) by the editors; DOI: 10.1002/adsc.201500316
(9) Kaushik Panigrahi, Gregory A. Applegate, Guillaume Malik, David B. Berkowitz* “Combining aClostridial Enzyme Exhibiting Unusual Active Site Plasticity with a Remarkably Facile Sigmatropic Rearrangement: Rapid Stereocontrolled Entry into Densely Functionalized Fluorinated Phosphonates for Chemical Biology. J. Am. Chem. Soc, 2015, 137, 3600-3609. DOI: 10.1021/jacs.5b00022; featured as a JACS Spotlight: DOI: 10.1021/jacs.5b02757
(10) Christopher D. McCune, Matthew L. Beio, Jacob A. Friest, Sandeep Ginotra, David B. Berkowitz* “A Useful Methoxyvinyl Cation Equivalent: a-t-Butyldimethylsilyl-a-methoxyacetaldehyde” Tetrahedron Lett. 2015, 56, 3575-3579 (Special Symposium-In-Print honoring Harry H. Wasserman) DOI: 10.1016/j.tetlet.2015.02.122
(11) Xiang Fei, Thomas Holmes, Julianna Diddle, Lauren Hintz, Dan Delaney, Alex Stock, Danielle Renner, Molly McDevitt, David B. Berkowitz, Juliane K. Soukup “Phosphatase-Inert Glucosamine 6-Phosphate Mimics Serve as Actuators of the glmS Riboswitch” ACS Chemical Biology, 2014, 9;2875-2882; DOI: 10.1021/cb500458f